Do you have SI Joint Pain?
The SI joint can contribute to a large amount of lower back pain. The SI joint has been identified as a cause of discomfort in 15-30% of persistent lower back pain sufferers. Furthermore, the SI joint is involved in up to 43% of persons who have had a lumbar fusion and subsequently experience continued or new onset lower back pain.
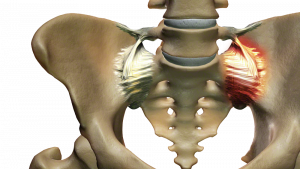
Excessive motion in the SI joint may inflame and disrupt the joint and surrounding nerves causing pain in the lower back, hip, groin, or pelvis.
Sacroiliac Joint (SI Joint) Anatomy
The sacroiliac (SI) joint is where the iliac bones of the pelvis connect to the sacrum, which forms the bottom part of the spine above the tailbone. The SI joint transfers energy between a person’s legs and torso.
The SI joint is prone to injuries and deterioration, which can bring discomforts in the vicinity of it, such as the lower back, hips, and legs. When putting pressure on the joints, this is particularly apparent when doing activities like lifting or jogging.
Pain from the SI joint can resemble a disc or low back discomfort, or it might be localized to the hip or groin. As a result, symptoms of an SI joint problem should always be considered in diagnosing lower back, hip, and pelvic pain.
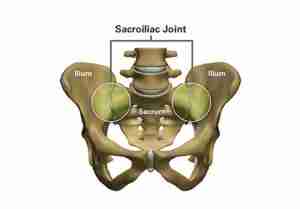
The SI joint is the link between the ilium in the pelvis to the sacrum, which is the lowest part of the spine above the tailbone.
Making a Diagnosis
Physical examination may result in the detection of the SI joint as a source of your symptoms. Due to their ability to rule out other typical sources of pain, X-rays, CT scans, and MRI scans may be useful in determining if an SI joint issue is present. It’s also worth noting that other problems (such as a disc issue) can sometimes occur alongside SI joint abnormalities.
The most reliable technique to determine whether the SI joint is the source of your lower back discomfort is to anesthetize it with a local anesthetic. To verify correct needle placement in the SI joint, this diagnostic injection will be given under X-ray or CT guidance. If your pain improves by at least 50%, it can be assumed that the SI joint is either the source of or a significant contributor to your lower back, hip, or pelvic discomfort. It’s less probable that the SI joint is responsible for your suffering if the pain does not improve after injection into the SI joint.
If you have any of the following symptoms, you may have pain in the sacroiliac joint.

SI joint pain can mimic pain in the lower back, hip, groin, or pelvis.

Patients who suffer from SI joint dysfunction can have severe pain when performing transitional movements like standing from a chair.

Patients who have SI joint pain usually find it difficult to sit for long periods of time, and usually try to alleviate the discomfort by sitting on the least effected side.
Indications
The iFuse Implant System® is a procedure that fuses the sacroiliac joints together to treat sacroiliac joint dysfunction caused by deterioration or disruption of the sacroiliac joint. This includes symptoms that began during pregnancy or in the peripartum period and have persisted beyond 6 months after delivery. This product is also helpful for patients who need sacroiliac fusion to improve immobilization and stabilization of the sacroiliac joint. It works well as part of a lumbar or thoracolumbar fusion, or for acute, non-acute, and non-traumatic fractures involving the sacroiliac joint. There are potential risks associated with the iFuse Implant System. It may not be appropriate for all patients and all patients may not benefit. For information about the risks, visit www.si-bone.com/risks
Treatment Options
If your doctor confirms that your SI joint is the source of discomfort, they will begin treatment options. Some people see significant improvement from physical therapy, SI belt wear, or taking an oral medication, however, these solutions are not always permanent. Surgery may be an option if you have tried other methods without finding long-term relief – including the minimally invasive iFuse procedure.
SI Joint Fusion with the iFuse Implant System®
The iFuse Implant System is meant to stabilize and fuse the SI joint. To improve SI joint stability, reduce pain, and increase function, the iFuse technique involves inserting triangular-shaped titanium implants across the sacroiliac joint. The operation takes about an hour via a tiny one-inch incision. The Triangular Design of the iFuse Implants has been clinically evaluated more than any other SI joint fusion surgery.
Dr. Morton has extensively trained in minimally invasive surgical (MIS) techniques, including the iFuse Implant System® from SI-BONE®, a medical device company specializing in MIS sacroiliac (SI) joint treatment. The iFuse implant is backed by over 100 peer-reviewed publications detailing its safety, long-term effectiveness, and biomechanical/economic benefits (www.si-bone.com/results). Furthermore, the iFuse implant is the only SI joint fusion device supported by multiple prospective clinical studies–including two randomized controlled trials affirming that treatment with the iFuse improves pain, patient function, and quality of life. The iFuse Implant System, like any minimally invasive surgical treatment, comes with a number of possible hazards. It may not be suitable for some patients or everyone may not profit from it. Visit www.si-bone.com/risks for more information about the dangers involved with this surgery.
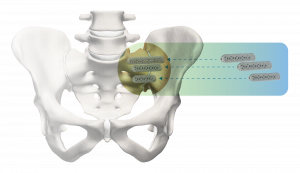
The iFuse Implant System is designed to provide stabilization and fusion for certain SI joint disorders.
References
1. Bernard TN, et al. Recognizing specific characteristics of nonspecific low back pain. Clin Orthop Relat Res. 1987;217:266–80.
2. Schwarzer AC, et al. The Sacroiliac Joint in Chronic Low Back Pain. Spine. 1995;20:31–7.
3. Maigne JY, et al. Results of Sacroiliac Joint Double Block and Value of Sacroiliac Pain Provocation Tests in 54 Patients with Low Back Pain. Spine. 1996;21:1889–92.
4. Sembrano JN, et al. How Often is Low Back Pain Not Coming From The Back? Spine. 2009;34:E27–32.
5. DePalma MJ, et al. Etiology of Chronic Low Back Pain Patients Having Undergone Lumbar Fusion. Pain Med. 2011;12:732-9.
6. Polly DW, et al., and the INSITE Study Group. Two-Year Outcomes from a Randomized Controlled Trial of Minimally Invasive Sacroiliac Joint Fusion vs. Non-Surgical Management for Sacroiliac Joint Dysfunction. Int J Spine Surg. 2016;10:Article 28. DOI: 10.14444/3028
7. Dengler J, et al. Randomized Trial of Sacroiliac Joint Fusion vs. Conservative Management for Chronic Low Back Pain Attributed to the Sacroiliac Joint. J Bone Joint Surg Am. 2019;101(5):400-11. DOI: 10.2106/JBJS.18.00022.
8. Duhon B, Bitan F, Lockstadt H, Kovalsky D, Cher D, Hillen T, on behalf of the SIFI Study Group. Triangular Titanium Implants for Minimally Invasive Sacroiliac Joint Fusion: 2-Year Follow-Up from a Prospective Multicenter Trial. Int J Spine Surg. 2016;10:Article 13. DOI: 10.14444/3013
9. Dengler J, et al. on behalf of the INSITE, iMIA and SIFI study groups. Predictors of Outcome in Conservative and Minimally Invasive Surgical Management of Pain Originating from the Sacroiliac Joint – a Pooled Analysis. Spine. 2017;42(21):1664-73. [Epub 2017 Mar 27]. DOI: 10.1097/BRS.0000000000002169
10. Whang PG, et al. Long-Term Prospective Clinical and Radiographic Outcomes After Minimally Invasive Lateral Transiliac Sacroiliac Joint Fusion Using Triangular Titanium Implants. Med Devices (Auckl). 2019;12:411-422. DOI: 10.2147/MDER.S219862
11. Patel V, et al. Prospective Trial of Sacroiliac Joint Fusion Using 3D-Printed Triangular Titanium Implants: 24-Month Follow-Up. Med Devices (Auckl). 2021;14:211-216. DOI: 10.2147/MDER.S314828




